 At HMR, we’ve 2 state-of-the art sleep units and can do a wide range of sleep trials. In the past decade, our many sleep trials of novel compounds have given us experience of a wide range of measures of sleep performance –
At HMR, we’ve 2 state-of-the art sleep units and can do a wide range of sleep trials. In the past decade, our many sleep trials of novel compounds have given us experience of a wide range of measures of sleep performance –
- polysomnography
- actigraphy
- neurovigil
- EEG
- sleep and worry VAS
- Leeds sleep evaluation questionnaire
- HDRS17 Sleep Item-Adjusted
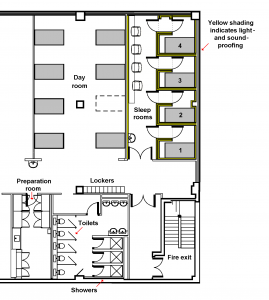
Our 2 sleep units each comprise 4 bedrooms, some with an en-suite toilet and shower. Each room has its own entrance lobby, to maintain sound- and light-proofing when subjects or HMR staff enter the room. The units have the highest level of soundproofing, with acoustic board, acoustic tiles, and 10 cm thick fibre insulation in walls and ceiling. Other features include
- remote-controlled air conditioning and ventilation
- mains and low-voltage cables in separate trunking channels
- shielded mains cables, and wooden beds, to prevent electromagnetic interference
- infrared light and video camera
- controlled light for <5 lux waking environment
- microphone
- external port for remote intravenous blood sampling
- Embla N7000 polysomnography (PSG) system with REMbrandt, including: EEG, EOG, ECG, and EMG electrodes and leads; oximeter; and sensors for respiratory effort, airflow, body position, nasal cannula, thermistor, and snoring.
- PSG equipment validated in accord with 21 CFR part 11.
Our 6 acquisition PCs are each protected by UPS, and store data on mirrored RAID drives. We copy the data across the network to our file server, which we back up daily. A PSG specialist reviews all data, as required for each trial.
We have extensive experience of phase 1 trials of novel CNS compounds; an MIA(IMP) to manufacture sterile and non-sterile products; and MHRA Phase 1 Supplementary Accreditation.

